How To: Create Double Step Kinetic Model for DSC Data
Curing of Epoxy Resin
Introduction
In this "How To:", a double step kinetic model for DSC data will be created. These data have two peaks of different directions:
- endothermal melting,
- exothermal curing.
This is the two-component curing system, where curing occurs immediately after melting of one component in this system.
We will start by loading a sample data project included in Kinetics Neo, will then create a kinetics model consisting of only one main step, will then add the small additional step to have double-step model.
Just a few clicks in over the course of a few minutes - and you will have your kinetics model!
Sample data:
- Data type: differential scanning calorimetry (DSC)
- Project file: Ep_Resin_DSC_Data.kinx
Load the Sample Data Project
1. Start the Kinetics Neo software.
Click on the blue "File" tab to open the application menu.
2. Open Sample Data DSC project.
Click on "Open" in the menu on the left side, then select "Samples". The Kinetics Neo samples directory will be opened in Windows Explorer.
Select directory "DSC_Ep_Resin".
3. Open the Kinetics Neo project file "Ep_Resin_DSC_Data.kinx" .
Check the Loaded Measurement Data
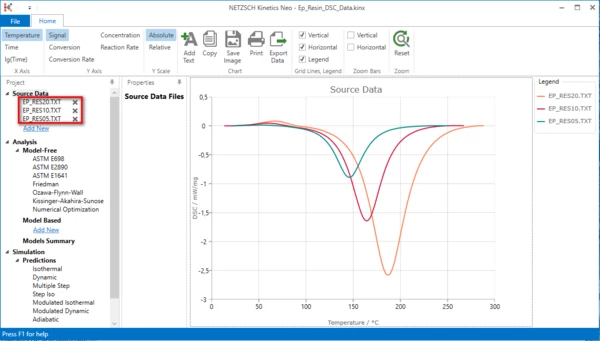
4. Check whether the DSC measurement data are loaded.
The Kinetics Neo sample project "Ep_Resin_DSC_Data.kinx" already contains imported sample DSC data files for Curing of Epoxy Resin:
- Ep_RES20.TXT – heating rate 20K/min
- Ep_RES10.TXT – heating rate 10K/min
- Ep_RES5.TXT – heating rate 5K/min
If the project file is successfully loaded then these file names will be seen in the "Source Data" section on the left side. The data curves will be shown on the main chart.
Create a One-Step Kinetic Model for Exothermal Effect
5. Add new model: In "Analysis" tree under "Model Based" click on "Add New".
A new "Model Based" kinetic model will be created with default parameters:
- One step: A->B .
- Reaction type: F1, 1st order reaction.
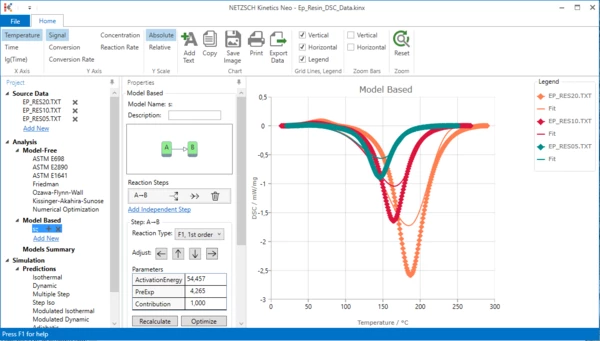
6. Change reaction type for A-B kinetic step
It is known that curing reactions are usually autocatalytical reactions.
In this case it is recommended to select reaction with autocatalysis "Cn" with unknown reaction order "n" and unknown order of autocatalysis: "Cn, n-th order with autocat.". Using model optimization the software will determine the correct reaction order on its own.
Select step A-B, then select reaction type "Cn, n-th order with autocat.".
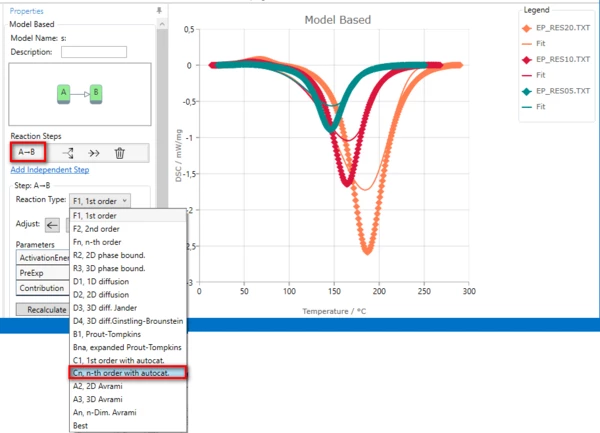
Result after changing of reaction type to Cn:

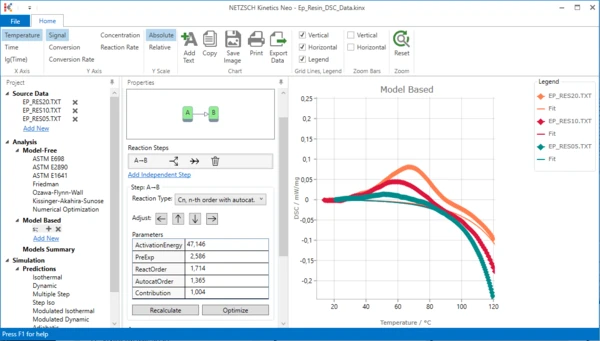
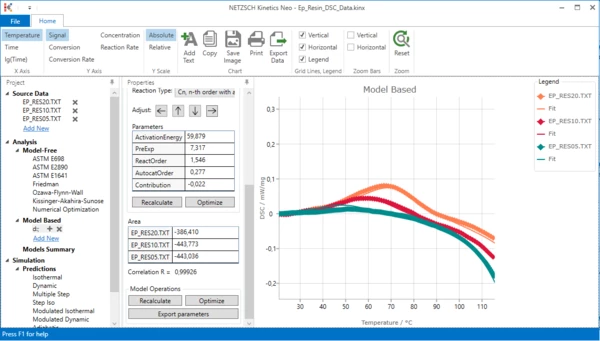
7. Optimize one-step model. In “Model Operation” section at the lower part of Properties panel select “Optimize”.
The model step will be optimized. This may take a few seconds...
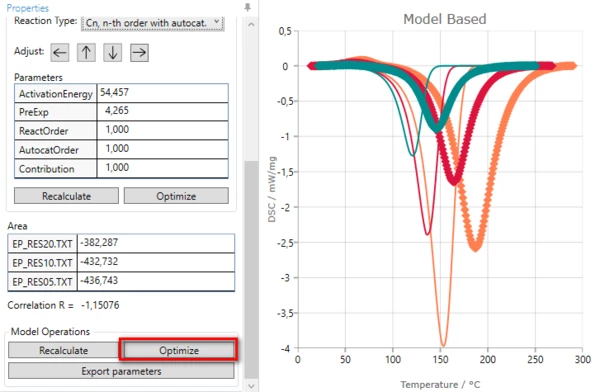
Result after model optimization:
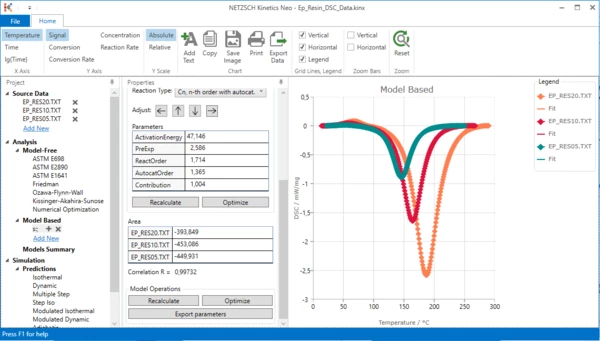
One-step kinetic model is ready. It describes the exothermal curing process. But is it enough to fit all data? Unfortunately not.
Please zoom in the top left area of the chart: move mouse to the desired area, hold left mouse button and select area to zoom. You will see that there are significant differences between measurement and simulated data. This is because our 1-step kinetic model can't describe the endothermal melting peak at 50°C - 100°C.
In top Ribbon toolbar click Reset to set chart zoom to original 100% and see the complete chart.
Extend the Kinetic Model by Adding of a New Step for Endothermal Effect
We would like to add the additional step, which has the meaning of endothermal melting at 50°C - 100°C. It is known from the chemistry of the process, that the curing occurs after melting. Thus, we would like to create double-step kinetic model, where the steps are connected consequently.
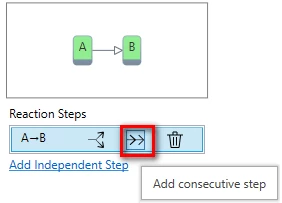
8. Add consecutive step.
In Properties panel select step A-B, and click on “Add consecutive step” symbol.
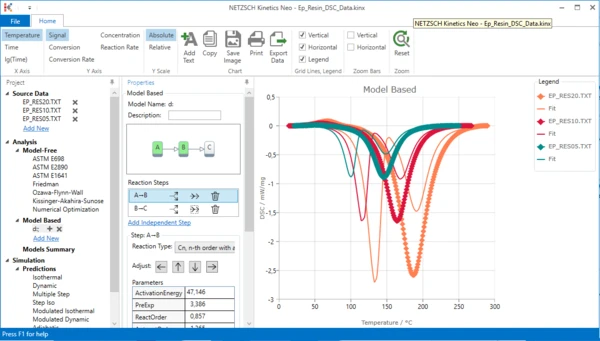
Now the simulated model has two steps, but the first of them is too late and still exothermal. We should make it endothermal and then move to the lower temperatures.
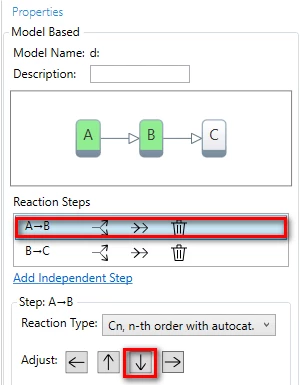
9. Change step direction
In the Properties panel in the list of Reaction Steps select the first step A-B and then reduce its contribution (click on arrow down).
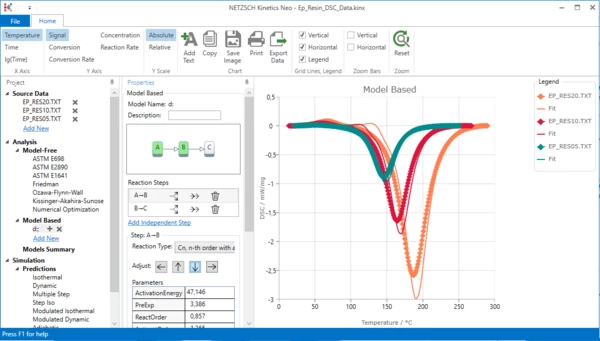
10. Move the first step to the left.
Select step A-B and click arrow left.
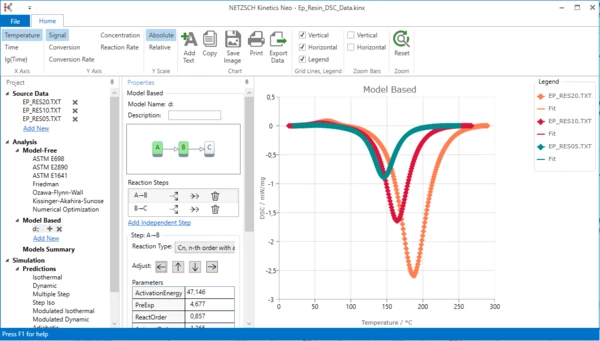
11. Zoom endothermal peak: select the zoom area by the mouse by holding the left mouse button.
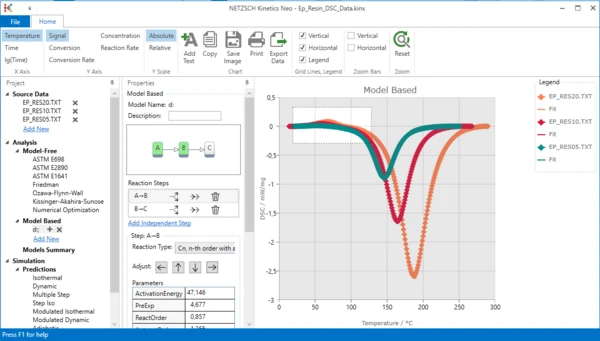
The chart range is zoomed:
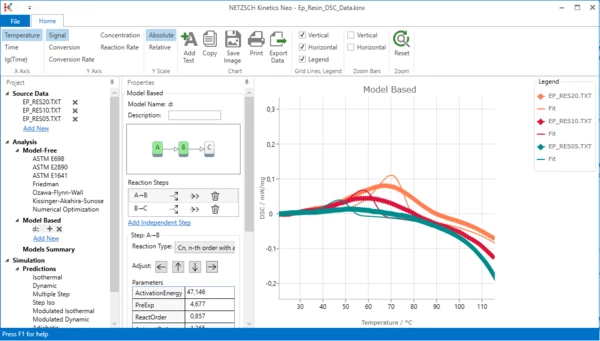
The endothermal peak is placed to the correct position, but its parameters are not optimal.
Optimize Model
12. Optimize model: in Model Operations select Optimize.
Now the endothermal peak has the optimal parameters.
In the top ribbon toolbar click Reset to set chart zoom to original 100% and see the complete model:
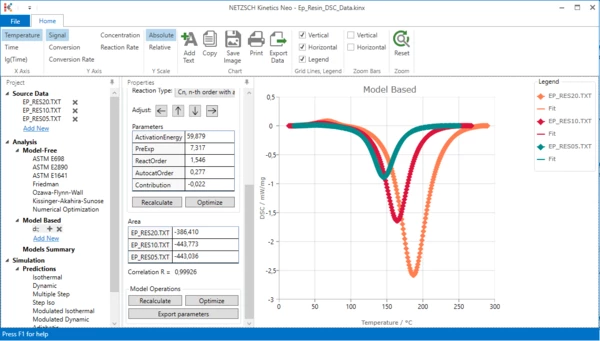
Now two-step kinetic model with endothermal and exothermal steps is ready.
